The Gansons Wash-In-Place (WIP)/(CIP) System
We have engineered a fully automated wash-in-place system specifically for pharmaceutical manufacturing, ensuring rigorous cleanliness and eliminating risks of cross-contamination. Tailored to each facility’s layout and equipment configuration, this system delivers validated hygiene, regulatory compliance, and minimal downtime for maximum safety and productivity.
Granulation Lines in Sun Pharma
0
+
Kg of Tablet Produced everyday
+

Smart Automation & Unmatched Cleaning Precision
- Fully Automated with Multiple Configurations: Gansons Wash-in-Place (WIP) system is designed for seamless automation, eliminating manual intervention. Its flexible configurations adapt to diverse production environments, ensuring optimal cleaning performance for tanks, pipelines, and equipment of varying sizes and complexities.
- Internal Duct Washing with Automated Spray Nozzles: The integrated duct washing system uses programmable spray nozzles to deliver high-impact, 360° coverage. These nozzles adjust pressure and flow dynamically, ensuring thorough residue removal from intricate ductwork and hard-to-reach areas.
Smart Heating & Accurate Chemical Dosing
- Inline Heating for Enhanced Cleaning Equipped with inline heating, the system maintains precise water temperatures to boost detergent efficacy. This feature accelerates cleaning cycles, ensures sanitization, and reduces energy consumption through smart heat management.
- Intelligent Detergent Dosing System: An automated dosing mechanism injects detergents and sanitizers with accuracy, optimizing chemical usage. This reduces waste, lowers operational costs, and ensures consistent cleaning outcomes without over- or under-dosing risks.


Smart Heating & Accurate Chemical Dosing
- Inline Heating for Enhanced Cleaning Equipped with inline heating, the system maintains precise water temperatures to boost detergent efficacy. This feature accelerates cleaning cycles, ensures sanitization, and reduces energy consumption through smart heat management.
- Intelligent Detergent Dosing System: An automated dosing mechanism injects detergents and sanitizers with accuracy, optimizing chemical usage. This reduces waste, lowers operational costs, and ensures consistent cleaning outcomes without over- or under-dosing risks.
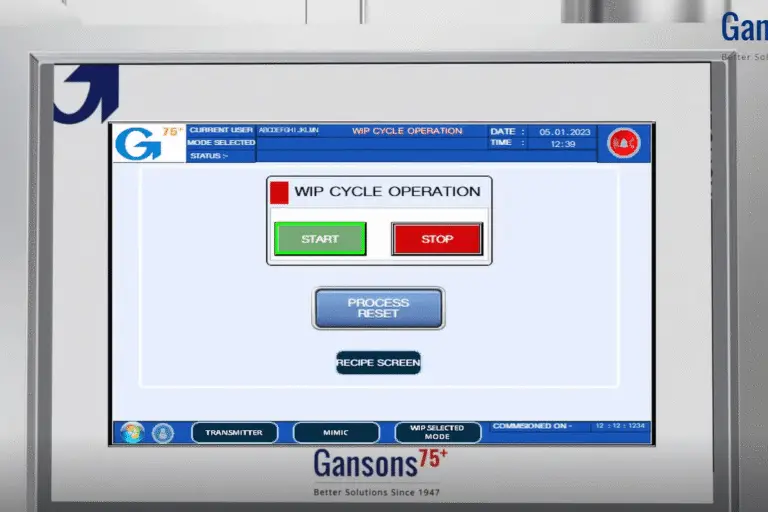
A standardized cleaning approach
- Fully Recipe-Driven Operation: The system operates via customizable digital recipes, allowing users to preset parameters like cycle duration, temperature, and chemical concentration. This guarantees repeatability, compliance, and effortless adaptation to different product lines or hygiene standards.
- Deionized Water Integration: A dedicated deionized (DI) water provision prevents mineral buildup and ensures spotless rinsing. This is critical for industries requiring ultra-pure standards, such as pharmaceuticals, biotechnology, and high-precision manufacturing.

Automation and Control
The Gansons automation platform is designed for pharmaceutical regulatory compliance at every level – from batch record generation to hazardous area operation:
- 21 CFR Part 11 compliance - electronic batch records, immutable audit trail, electronic signatures
- ALCOA+ data integrity - all process data attributable, contemporaneous, and audit-ready
- ATEX rated and compliant as per client configuration
- IoT enabled - real-time process analytics, remote monitoring, condition-based maintenance
- Pharma 4.0 ready - smart graphs for lot reproducibility, MES integration via OPC-UA
- Three-mode operation - with multi-level password access and role-based operator control
Installed in facilities regulated by US-FDA, UK-MHRA, EMA, WHO-GMP, Japan-PMDA, TGA, SAHPRA, MCC, and health authorities in 40+ countries. CE, UL, ATEX, ISO, and IEC configurations available on request.
Compliance and Certifications







